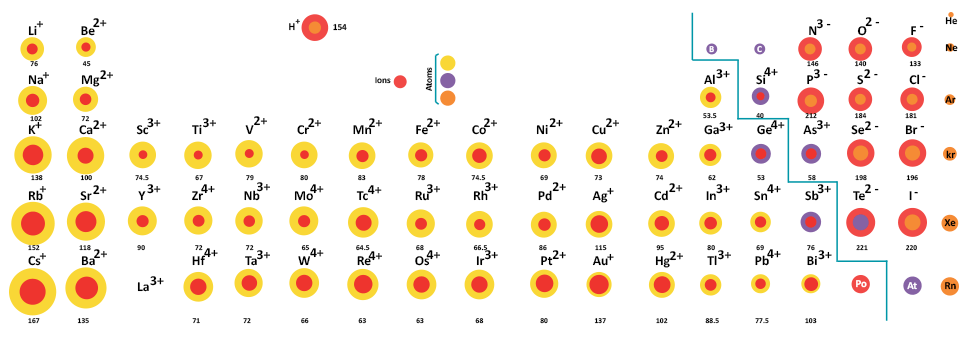
Because of their proximity to the nucleus, s-orbital electrons penetrate and shield more effectively than p-orbital electrons within the same energy level (represented by the Principal quantum number, n), while p electrons penetrate and shield more effectively than d-orbital electrons. Shielding and penetration are affected by changes in orbital Penetration is the existence of an electron inside an inner electron’s shell, while shielding is the process by which an inner electron shields an outer electron from the nucleus’ full attractive force, lowering ( Z eff ). Shielding and PenetrationĮlectromagnetic interactions between electrons in an atom change each electron’s effective nuclear charge ( Z eff ). Because the number of electrons exceeds the number of protons, the ionic radius of nonmetals increases as the effective nuclear charge falls. The ionic radius of metals generating cations diminishes as you progress through a row of the periodic table, as the metals lose their outer electron orbitals. It may appear odd that the size of an ion shrinks when additional protons, neutrons, and electrons are added over time. Why does the radius of a group rise as the atomic number increases? Additional layers of electrons are added as you progress down a group in the periodic table, causing the ionic radius to naturally rise as you move down the periodic table. The ionic radius is comparable to but not identical to the atomic radius, because the ionic size is determined by the distribution of its outermost electrons and is inversely proportional to the ionic effective nuclear charge. The size of a spherical ion is measured by the ionic radius of the ion (ion) of an atom (either a cation or anion). Radius decreases as you walk down a group and increases as you move across a period (row) (column).Īn ion is an atom that has acquired an electrical charge by gaining or losing one or more electrons. Atomic and ionic radius follow the same pattern on the periodic table.This number might be the same as the atomic radius, or it could be greater for anions and less for cations, or it could be the same for both. The ionic radius is equal to half the distance between two interacting gas atoms.In other words, it is the diameter of half an atom across the outer stable electrons. The atomic radius of a neutral atom is half its diameter.Atomic radius, ionic radius, covalent radius, and van der Waals radius are all distinct ways to quantify the size of an atom.Negative ions have larger atoms than their neutral counterparts. The size of positive ions is less than that of corresponding uncharged atoms. The ionic radius of an element differs from its atomic radius. The anions are formed by adding two electrons per atom to the group 16 elements and one electron per atom to the group 17 elements. It’s worth noting that the ions in group 16 are larger than those in group 17. Because there are more electrons, there are more electron-electron repulsions, which causes the electron cloud to spread out. When the electrons outnumber the protons, the protons’ total attractive attraction for the electrons decreases. 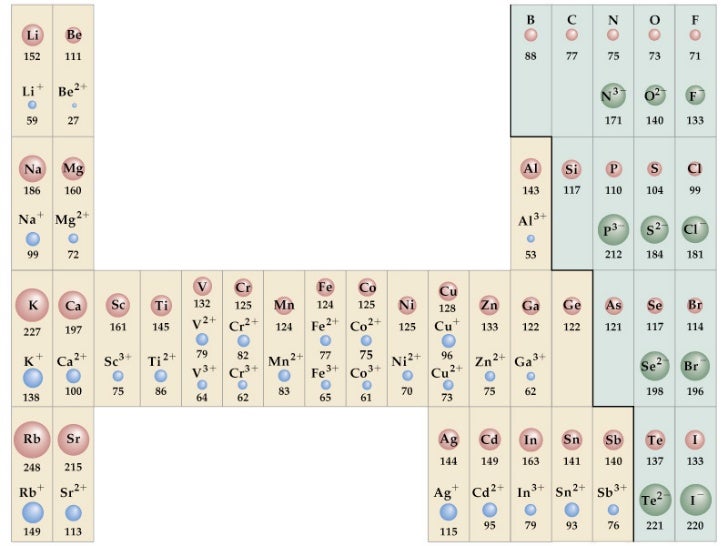
When electrons are added to a parent atom, the result is always an anion that is larger than the parent atom. To make the equivalent ion, one electron is lost from the potassium atom, while calcium loses two electrons. The number of electrons ejected is also a consideration. Another reason is that because protons now outweigh electrons, the remaining electrons are drawn closer to the nucleus. The valence electron(s) are eliminated, and the resultant ion has one fewer occupied primary energy level, resulting in a smaller electron cloud. When electrons are removed from an atom, the result is always a cation that is much smaller than the parent atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
